Production of Power with Next-Gen Clean Energy Technology
“DME is the silver bullet solution to utilize existing vehicular technology and can be easily adopted in diesel vehicles, petrol cars, auto rickshaws, scooters, and motorcycles. When used universally, it has the potential to completely eliminate vehicular pollution in cities like Delhi.”
In Brief
- DME, a colorless and chemically stable gas, has shown promise as a fuel for diesel engines. Blending DME with diesel can reduce pollution, improve mileage, and emit less soot and carbon dioxide. DME’s unique properties enable soot-free combustion, making it a cleaner substitute for diesel vehicles.
- DME can be produced through direct or indirect processes using syngas derived from natural gas or renewable sources like biomass, solar, and wind.
- Additionally, DME production from CO2 offers a promising pathway for carbon dioxide valorization. Ongoing research focuses on developing catalysts and membranes to optimize DME production from CO2-rich feedstock while reducing greenhouse gas emissions.
Production of Di Methyl Ether or DME: A Clean Fuel
DME is a synthetic fuel that possesses compatible properties, such as a high cetane number and low auto-ignition temperature. Despite its easier evaporation and lower density, DME’s spray characteristics enable its application in compression-ignition (CI) engines. Its simple chemical structure (CH3-OCH3) with only C-H and C-O bonds, and no C-C bond, along with its high oxygen content, result in soot-free combustion. These factors justify considering DME for use in diesel engines. When blended with diesel, DME-Diesel blend has a heat value of 39 MJ compared to 42 MJ for diesel alone. Diesel vehicles emit 25 to 400 times more particulate black carbon and associated organic matter (“soot”) per kilometer when using neat diesel. However, when DME is blended with diesel, it results in zero particulate matter emissions (SPM).
Direct and Indirect production processes
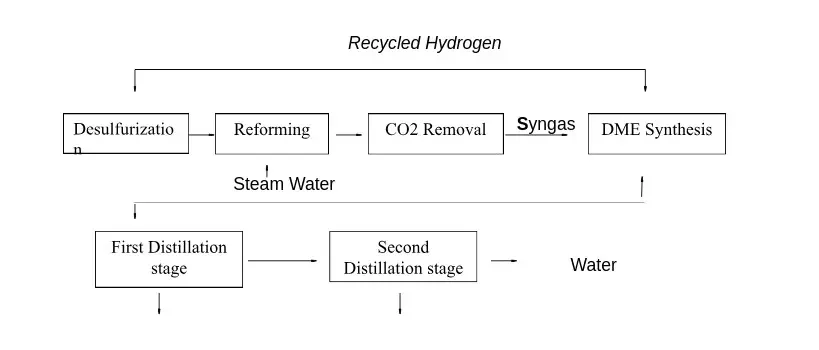
In industrial applications, DME is produced from syngas through two different configurations: the one-step process and the two-step process. In the one-step process (direct production process), DME is directly produced from syngas in a single reactor where a bi-functional catalyst supports both methanol formation and methanol dehydration, as described in the following reaction scheme:
Methanol formation: CO + 2H2 ↔ CH3OH ΔHo = -90.4 kJ/mol
Water-gas shift: CO + H2O ↔ CO2 + H2 ΔHo = -41.0 kJ/mol
Methanol dehydration: 2CH3OH ↔ CH3OCH3 + H2O ΔHo = -23.0 kJ/mol
Overall reaction: 3CO + 3H2 ↔ CH3OCH3 + CO2 ΔHo = -245 kJ/mol
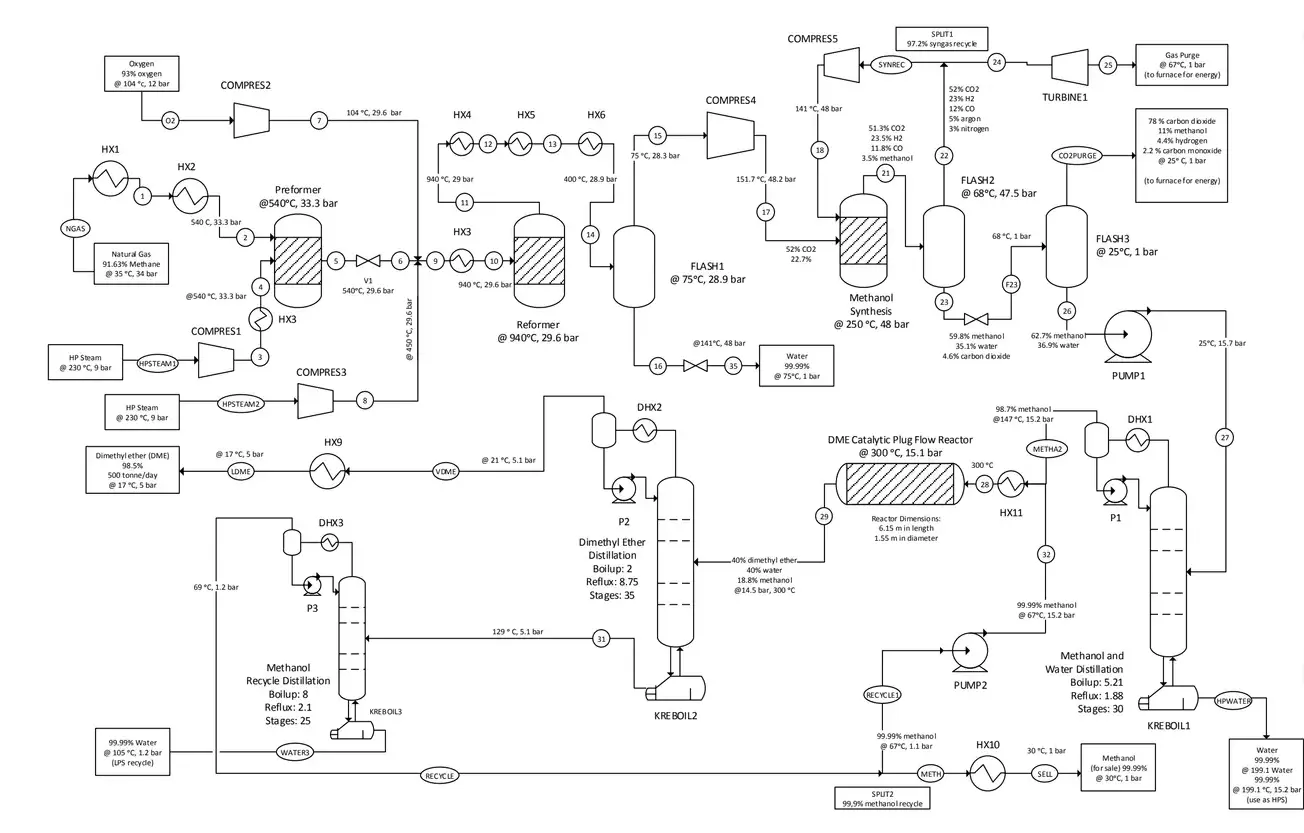
The syngas is produced through natural gas steam reforming or coal/petroleum residues gasification. After the DME synthesis reactor, a purification unit is required to separate DME from water and methanol in a double distillation stage. The following figure illustrates a diagram of the one-step process.
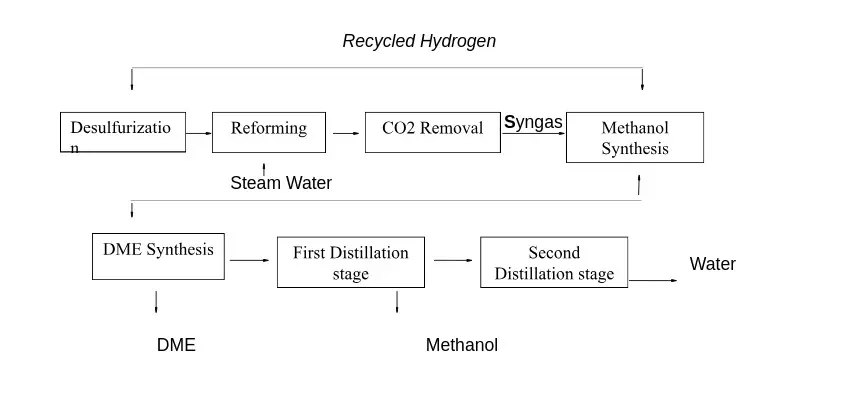
In the two steps (indirect) process, the methanol formation from syngas and the DME production from methanol are supported in two separated reactors, where the specific catalysts (copper-based for the first, silica-alumina for the second) are packed. The figure illustrates the block diagram of this architecture.
DME production from renewable energies
DME production from renewable energies he reactants of the DME synthesis process can be produced from renewable energy as biomass, solar and wind. By this way, the DME is a sort of liquid energy vector, able to store the renewable energy in a easily dispensable, easy applicable and high-energy density fuel. Starting from biomasses as energy crops, agro-residue, forest residue, etc., a gasification process can be applied to generate a syngas stream to be fed to one-step or two-steps DME synthesis process. On the other hand, if the starting biomass is an organic trash, manure or sewage, an an aero bio digestion + pyrolysis system can be applied to generate the CO and H2 stream.
The hydrogen stream in the syngas mixture can be generated by an electrolyzer supplied by electricity produced from renewable power plants as photovoltaics and wind farms and then mixed with CO/CO2. By this way, the renewable energy is “stored” in the DME, which, being a liquid fuel, can be easily distributed, stored and used, differently from the hydrogen itself which has a series of unsolved issues related to the distribution and storage. The following scheme shows a conceptual layout of the DME production from solar/biomass energy.
DME production as a CO2 valorisation process
DME production as a CO2 valorisation process Instead of the syngas, a CO2-rich feedstock can be supplied to the DME production process, thus converting the CO2 in a high added value product. By this process, the CO2, which is the main Green House gas (GHG), is not emitted but is converted into a fuel which can be burned releasing again the carbon dioxide.
Such a configuration is less developed than the conventional syngas-fuelled process, but many research efforts are devoted to improve its performance since it would allow both the production of DME and the reduction of GHG emissions, thus reducing the carbon footprint of DME synthesis.
CO2 presence in the reactor environment leads to two main issues:CO2-rich feedstock influences the active state of the catalyst for methanol synthesis, reducing the rate of formation of methanol
CO2 promotes the reverse Water Gas shift reaction, thus producing H2O and inhibiting the methanol dehydration.
The research is focused mainly on the development of new catalyst, tailored for CO2-rich mixture conversion, and of selective membranes able to remove water from the reaction environment, promoting the methanol dehydration reaction and the DME production.